
Oct 2019 – It is with great pleasure to announce EyRIS’s first medical device after Heath Science Authority of Singapore (HSA) gave us the official approval. Our medical device, SELENA+, is an effective assistive tool in the diagnosis of 3 eye diseases, diabetic retinopathy (DR), Glaucoma and Age-Related Macular Degeneration (AMD).
This approval closely follows the ISO13485:2016 certification we received for our Quality Management System (QMS) where we had to demonstrate our ability to provide medical devices and related services that consistently meet customer and HSA regulatory requirements.
There are more than 415 million diabetics in the world today who require an annual eye check-up to prevent vision loss. Statistically, 3 in 10 diabetics suffer from diabetes induced eye disease and 1 in 10 develops blindness when left untreated.
In South-East Asia alone, there are near 100 million persons who suffer from diabetes, more than 10% of the population. A World Health Organization report in 2017 highlighted a recent dramatic increase in diabetes and critically, “half of those cases remain undiagnosed, underscoring the need for rapid, low-cost solutions to reach the region’s underserved areas”.
With the approval, we aim to assist stakeholders develop their screening programs & protocols, implementing their diabetes screening plans at the primary health-care level using SELENA+ grade fundus images cheaper and faster so that follow-up care is directed to where it is most needed.
About SELENA+
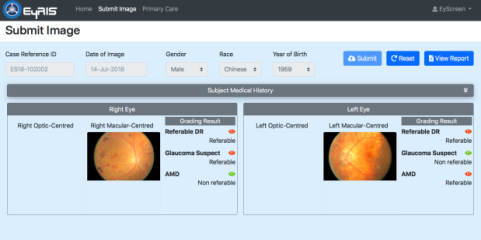 Early screening is highly effective in helping to prevent blindness from a range of eye diseases. SELENA+ is an intelligent deep learning system that can detect eye diseases: this system was jointly invented by researchers at NUS School of Computing (NUS-SoC) and the Singapore Eye Research Institute (SERI) to perform automated image analysis of retinal images. The analysis results will be used to provide early detection of eye diseases, including diabetic retinopathy (DR), glaucoma and age-related macular degeneration (AMD).
Early screening is highly effective in helping to prevent blindness from a range of eye diseases. SELENA+ is an intelligent deep learning system that can detect eye diseases: this system was jointly invented by researchers at NUS School of Computing (NUS-SoC) and the Singapore Eye Research Institute (SERI) to perform automated image analysis of retinal images. The analysis results will be used to provide early detection of eye diseases, including diabetic retinopathy (DR), glaucoma and age-related macular degeneration (AMD).
Comments are closed.

